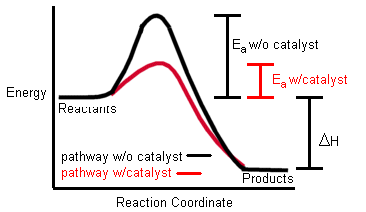
FACTORS THAT AFFECT RATES OF REACTION
The Kinetic Molecular Theory states that matter is made up of particles that are always in constant motion and that the particles move more quickly at higher temperatures. From this theory came the COLLISION MODEL for chemical reactions. According to the collision model there are two ways to make a chemical reaction go faster: 1) …