Onion Root Cell Cycle Lab Answers
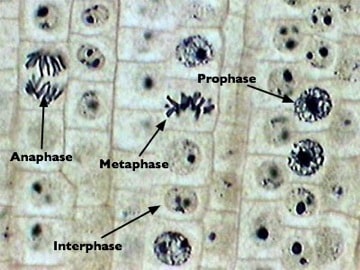
Introduction Every somatic cell undergoes a phase called mitosis. Mitosis is the division of the nucleus to form two genetically identical nuclei. There are four phases of mitosis: prophase, metaphase, anaphase and telophase. Prior to mitosis is interphase (when the cell grows and duplicates all organelles), and post-mitosis is cytokinesis (when the cell membrane pinches…