What is Anesthesia?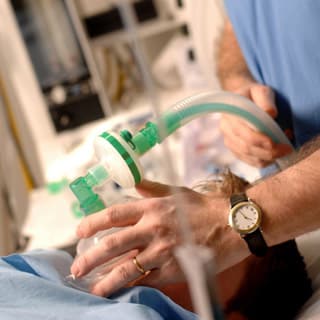
- A reversible lack of awareness caused by the inhalation (of gases) or injection of (liquid) drugs
- It is used to minimize pain, discomfort, and shock for surgical patients
- It can put medical staff at ease knowing that they won’t hurt the patient
General Anesthesia
- A loss of consciousness in the patient accomplished through injected and inhaled drugs
- Purpose: provides loss of pain response, loss of memory, loss of motor reflexes
- The anaesthetic affects the brain
- Used for lengthy surgeries or where the patient needs to be held in a specific position
Local Anesthesia
- A loss of sensation in a certain area of the body through injected drugs
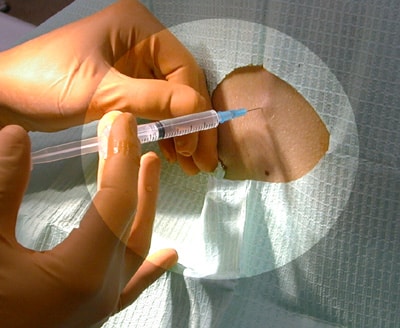
- Is achieved when the anaesthetic “freezes” the nerves around a specific area of the body
- Some common local anaesthetics are: procaine, bupivicaine and proparicaine
- Much safer that general anesthesia, as only one part of the body is receiving the drug
History of Anesthesia
- For most of history, all surgical procedures were performed without any effective painkillers
- Alcohol, opium, cocaine, blood letting, extreme cold and concussions were used as first anaesthetics
- In Canadian/American wars, solders had to get amputations without any painkillers
- Beginnings of Modern Anesthesia
- The first major breakthrough in anesthetics occurred on October 16 1846 when William T.G. Morton performed the first successful live demonstration of diethal ether anesthesia on a patient
- The anesthesia allowed Morton to painlessly remove a tumor from the back of his patients neck
- Ether was among the first of successful anesthetics to be used but it had several drawbacks: it induced vomiting and was very flammable
Evolution of Anesthesia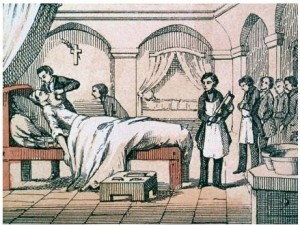
- Over time, anesthesia has improved and new chemicals that create an anesthetic effect have been discovered through trial and error
- These include nitrous oxide[N2O] Halothane [CF3CHClBr] and Isofluorine[CF3CHClOCHF2] (for general anesthesia)
A Good Anesthetic …
- Must be non toxic, with minimal side effects
- Must be non-flammable
- Must have a reasonably high blood (water) solubility to make sure it can be transported from the lungs to the brain (but not too high so the anaesthetic stays too long in the bloodstream)
- Has to have a high solubility in the fatty tissues of the brain so it will concentrate onto the cells of the nervous system to cause unconsciousness
Temperature, Pressure and Gas Solubility
- Gay Lussacs law states that Temperature and pressure are directly proportionate.
- The solubility of gas decreases with pressure (Henry’s Law)
- The solubility of gas decreases with temperature
- In anesthetics, the gas must have a boiling point slightly above room temperature so that the gas will be highly soluble
- Low temperature = low pressure
- However,the pressure of the gas cannot be too low because the gas needs a high enough pressure to enter the lungs
Disadvantages of Current Anesthetics
- The anesthetics we have today aren’t perfect
- Minor side effects can include nausea, shaking, extreme drowsiness for all types of anesthesia
- For general anesthesia, side effects can include heart attack, bruising, coordination problems, muscle pain, stroke, sharply falling blood pressure, and even death
