Purpose
In this lab we will observe the products of decomposition of potassium perchlorate (KClO4). We will then predict from our results the correct chemical reaction equation.
Procedure
1. Weigh out about 4.0g of KClO4 in a test tube. Record the accurate weight below.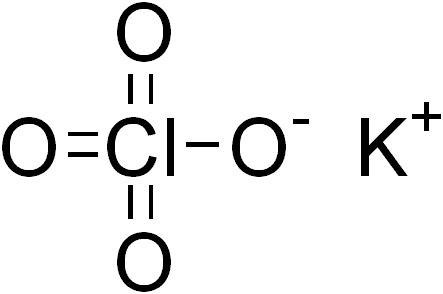
Product Weight Before Weight After
Mass of Test tube + KClO4 41.5g 39.8g
Mass of Test tube 37.5g 37.5g
Mass of KClO4 4.0g 2.3g
2. Set up the apparatus shown below.
3. Gently heat the test tube containing the potassium perchlorate. Gas should begin to collect in the collection bottle. Record all observation.
4. Once the reaction is complete, no more gas give off, allow the test tube to cool. While the test tube is cooling test the gas in the collection bottle with glowing splint.
Caution: Do not leave the rubber tubing down in the water trough during cooling or you will experience back-up.
5. After the test tube has cooled weigh it on a balance. What is the change in mass?
Observations
Oxygen flowed from the test tube into the bottle of water, forcing the water out.
Burning ember re-ignited when placed into the bottle of O2.
Calculations
1. The number of moles of KClO4 that we began with is .03 moles. 4.0g / 138.6g/mol = .03 moles
2. The number of moles of O2 that were present in our sample of KClO4 was .06 moles. 1.9g / 32g/mole = .06 moles
3. The number of moles of O2 lost is .02 moles. 1.7g / 32g/mol = .05 moles
4. KClO4 ->KCl + 202
4.0g / 138.6g/mol = .03 moles……………202 / KClO4 = .06 moles x 32g = 1.9g
5 Percent Yield: 89% O2 lost 1.7g ¸ O2 Expected 1.9g
