Surface Tension (N/m² = J/m³)
- Fluid property associated with the presence of a surface toward air
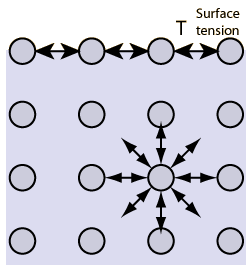
- Interfacial tension- is used to describe analogous phenomena for fluid having interfaces with solids or other liquids (can be +/-)
- Water molecules are forced toward the surface of a fluid due to placement on other molecules and attractive forces. (the attractive force at equilibrium in water but when pushed to the surface; attractive equilibrium is lost and pulls molecules away from the surface, but they can’t go anywhere because surface CAN’T shrink.
- This creates some internal pressure and forces liquid surfaces to contract to the minimal area.
- A force is required to hold the molecules at the surface area (Ơ) [high energy particles out the exterior with no “neighbor” molecules to hold it at equilibrium.]
- A fluid will shrink to the minimal surface area to maintain low energy
Ơ= ∆E/ ∆A (in J/m^2 = N/m=(kg/s^2) ***ALWAYS POSITIVE
W= ∆E = Ơ x ∆A
Ơ= F/lx
- WORK MUST BE DONE TO INCREASES SURFACE OF A FLUID (either force stretch a film or energy to increase surface area)
- Surface tension and pressure are the same (can be regarded as a FORCE, or ENERGY)
- Can be used on thin soap films due to its high cohesiveness
- Cannot use same set up for water; must place flat solid interface on it and determine the force needed to left solid off of the fluid.
Bubbles and Droplets
- Raindrops are an example of an open system
- Take the most geometrical shape to have the least energy to form surfaces
- Least energy/ surface area for a fixed volume…a SPHERE (alveoli take this shape)
- Energy is given off and raindrop ascertains sphere space;
Laplace Law
∆p= (4 x ơ)/ r (r= radius)
- Transmural pressure: ∆p= pinside-poutside (In lungs; the difference between alveolar & pleural pressure)
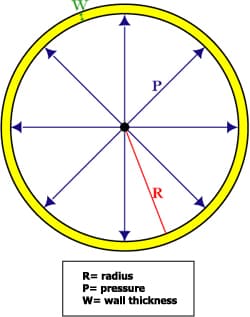
- The pressure inside the bubble is greater to stop it from imploding
- One Surface: Droplets, homogeneous cylinders
- Two Surfaces: Bubbles, hollow tubes
- Hollow/ Homogeneous tubes; finite curvature in only ONE direction across their surfaces
- Bubbles/ Droplets; finite curvature in only TWO directions across their surfaces
The pressure difference between the inside and outside of a fluid with a curved surface is INVERSELY proportional to the radius of curvature of the curved surface.
Smaller bubble, droplet, cylinder has a larger pressure difference ∆p
Formulas
∆p= pinside – poutside= (4 x ơ)/r [BUBBLES]
∆p= pinside – poutside= (2 x ơ)/r [HOLLOW CYLINDERS/ DROPLET]
∆p= pinside – poutside= (2 x ơ)/r [SOLID CYLINDERS]
***Surface tension for a capillary of small radius must be smaller than the surface tension of an arteriole with a larger radius:
rarteriole > rcapillary -> ơarteriole > ơcapillary
- Allows walls of capillaries to be thinner; this, in turn, improves the efficiency to be thinner; improves diffusion of O2 and transport of small ions [Small alveoli are more effective at gas exchange]
- Pulmonary Surfactants: wet the alveolar surface to counterbalance radius effect
- Neonatal Respiratory Distress Syndrome: premature baby can’t make surfactants and lung is stiff; alveoli collapse.
