Biological molecules are macromolecules, which basically means they are polymers composed on small repeating units that have been covalently bonded together. The exception to this is a type of lipid called triacyleglycerides or phospholipids which are relatively large molecules composed of smaller molecules bonded together.
Subunits are chemically liked together in the cell by a specific type of chemical reaction called a condensation reaction. These reactions are anabolic as they create larger molecules. When cells need to break molecules down (e.g. digestion), they perform catabolic reactions called hydrolysis.
CARBOHYDRATES
- Contain carbon, hydrogen and oxygen in a 1:2:1 ratio
- Contain multiple hydroxyl groups (How does this affect their ability to hydrogen bond?)
- Monosaccharides are simple sugars
- They can vary in their carbon chain length
- May assume a ring structure in water
- When glucose forms a ring there is a chance that the –OH group will be either above or below the plane of the of the ring
- Above the plane results in α-glucose
- They can vary in their carbonyl functional group
- Aldose – glyceraldehyde, ribose, glucose, galactose
- Ketose – dihydroxyacetone , ribulose, fructose
- Disaccharides are dimers (two monomers linked together)
- Common disaccharides include sucrose, maltose and lactose
- Monosaccharides liked by a glycosidic linkage.
Condensation Reaction/Dehydration Synthesis
LIPIDS
- hydrophobic molecules composed of C, H and O
- less –OH groups and longer hydrocarbons compared to carbohydrates therefore less soluble in H2O
- suited to their function of long term stored energy reserve
- and membrane components (barriers to water based cellular environment
four classes
- fats
- phospholipids
- steroids
- waxes
FATS
- common energy storage in animals within adipose tissue
- thermal insulation effect
- stored as triglycerides in plants and animals
- glycerol + 3 fatty acids
O
HO—C—CH2—CH2—CH3
O
HO—C—CH2—CH2—CH==CH—CH3
O
HO—C—CH2—CH2—CH3
Fatty acids can be either saturated (full number of hydrogen/no double bonds) or unsaturated (contain double bonds)
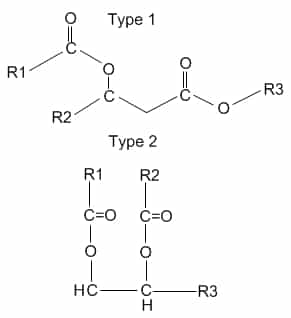
A condensation reaction occurs between the carboxyl group of the fatty acids and the hydroxyl groups of the glycerol (that’s why you need 3 fatty acids because there are 3 hydroxyl groups). The resulting bond is called an ester bond.
O O
R—OH + R1—C—OH -> R—O—C—R1 + H2O
The ester bond has limited hydrogen bonding capacity. In fact, fatty acids are stabilized by hydrophobic interactions. In a saturated fat the hydrophobic interactions are stronger because the fatty acid chains are able to stack very closely together, thus forming fats that are solids at room temperature. Unsaturated fats are liquids at room temperature because the double bonds in the chain allow for kinking in the chain which prevents close stacking of the molecules.
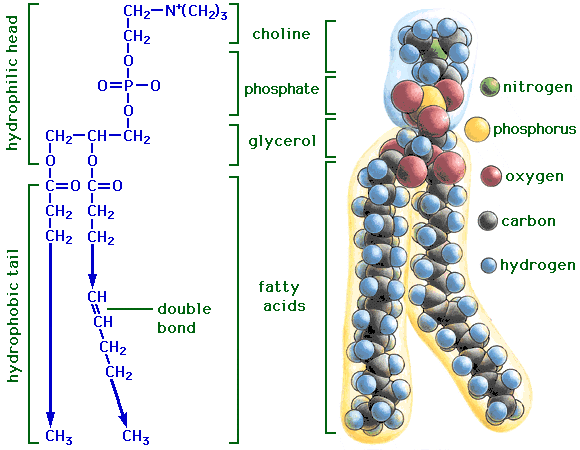
PHOSPHOLIPIDS
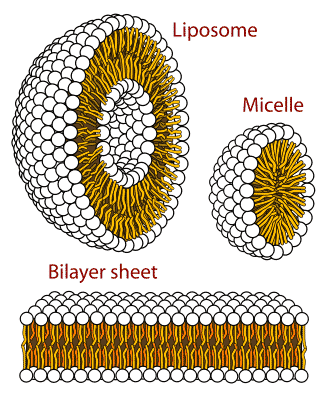
Phospholipids have a phosphate group on one of the carbons and only 2 fatty acids attached via an ester linkage. The presence of the phosphate group creates a hydrophilic end in this molecule and the fatty acid portion is hydrophobic. This makes the phospholipids s excellent barriers as in membrane structure.
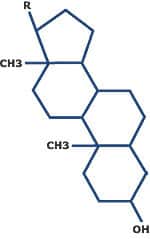
STEROIDS
- compact hydrophobic molecules containing four fused rings and various functional groups
- cells use steroids as a building block for certain vitamins like vitamin D
- some sex hormones are steroids
- cholesterol is an important membrane steroid that controls cell fluidity and permeability
WAXES
- hydrophobic molecules composed on long chain fatty acids linked to alcohols
- length of chain provides for pliable yet firm consistency
- form waterproof layers on plants


Dear schoolworkhelper
I am writing a book on applications of labs on chip to medical diagnosis and I would like too use your image of the lipid structure in the biochemistry chapter of the book.
I need your authorization to do so.
Please, if you would like to allow me the incorporation of your figure in the book, please fill the form below and send be back the filled form by e-mail.
Naturally I am available for comments and questions always via the above e-mail
Thanks for your kind attention.
Eugenio Iannone