Cornstarch: Upon adding 2 mL of cornstarch to the mixture, a visible change was observed. The mixture exhibited a significant increase in volume and took on a snow-like appearance, becoming more puffy. Stirring the mixture with a stirring rod gradually transformed the mixture’s color into a clear opaque white.
Sugar: The addition of 2 mL of sugar to the mixture caused a noticeable change. The sugar appeared to dissolve in the mixture, resulting in the formation of a jelly-like material. The color of the mixture became more translucent, shifting from its original opaque white appearance.
Salt: Upon adding 2 mL of salt to the mixture, distinct changes were observed. The mixture underwent a transformation, forming solid crystal-like particles. The color of the mixture remained opaque white, but its texture changed to a slush-like consistency, resembling crushed ice.
Parts of the Diaper:
Outer Back Sheet: The diaper’s outer back sheet is soft and thin, and serves to provide a soft touch upon the babies’ skin. Not only that but it also acts as a protective barrier which prevents liquid from leaking through the diaper.
Inside Top Sheet: The inside top sheet of the diaper is fluffy and substantially thicker than the outer sheet. The inside sheet serves for comfort and absorption of liquid, while keeping the baby’s skin dry and reducing the likelihood of irritation.
Adhesive Tape: The diaper’s adhesive tape allows for an easy fitting of the diaper on the baby and prevents the diaper from coming loose or shifting.
Elastic around leg cuffs: The elastic around leg cuffs serve to provide a secure fit against the babies legs while allowing stretch and comfort for the infant, and allowing them to move freely.
Fluffy Filling: The fluffy filling inside the diaper resembles cotton and provides absorbance of liquid, preventing the likelihood of irritation or diaper rashes. It also makes the diaper comfortable against the baby’s skin, providing a sort of cushiony feel for the baby.
ANALYSIS
Based upon the structure of sodium polymethacrylate and your knowledge of intermolecular forces, polarity, and organic chemistry, explain the changes observed to the crystals when water was added.
The structure of sodium polymethacrylate includes polar carbonyl groups (-C=O). Due to the electronegative oxygen atom and the partly positively charged hydrogen atoms in water, a polar molecule, hydrogen bonds can form. The interactions between the polymer’s carbonyl groups and the water molecules are made possible by the sodium polymethacrylate’s polar character.
The polar water molecules that are introduced to the sodium polymethacrylate crystals are drawn to the carbonyl groups of the polymer by hydrogen bonding. These hydrogen bonds cause the polymer’s crystal lattice structure to break down, which causes the polymer chains to hydrate. The hydration layer that the water molecules form around the polymer chains widens the space between them. As a result of the hydration process, the crystals of sodium polymethacrylate may undergo swelling or dissolution. The presence of water between the polymer chains causes the crystals to expand or swell as the chains become more flexible.
In some cases, the hydrogen bonds between the water molecules and the polymer can be strong enough to break the crystal lattice, resulting in the dissolution of the crystals and the formation of a homogeneous solution. The extent of the changes observed upon adding water depends on various factors, including the concentration of sodium polymethacrylate, temperature, and the amount of water added. Higher concentrations of the polymer and increased temperature can enhance the interaction between the water molecules and the polymer, leading to more pronounced swelling or dissolution of the crystals.
Based upon the structure of sodium polymethacrylate and your knowledge of intermolecular forces, polarity, and organic chemistry, explain the changes seen to the mixture with the addition of sugar, sodium chloride and baby powder.
Sugar: Sugar, including sucrose, contains many hydroxyl (-OH) groups, which make it a polar substance. With other polar molecules, these hydroxyl groups are capable of forming hydrogen bonds. The hydroxyl groups of sugar can interact with the carbonyl groups of the polymer in the case of sodium polymethacrylate, which has polar carbonyl groups (-C=O) in its structure. As a result of this interaction, hydrogen bonds are formed between the sugar molecules and the polymer chains, causing the mixture to appear like a network. This network gives the combination gel-like characteristics that raise its viscosity and give it a more semi-solid consistency.
Sodium Chloride: Sodium chloride (NaCl) is an ionic compound made up of positively charged sodium ions (Na+) and negatively charged chloride ions (Cl-). The sodium ions connected to the polymer backbone make up sodium polymethacrylate.
The chloride ions in the mixture may interact with the sodium ions in the polymer if sodium chloride is introduced. The ionic balance inside the polymer network may be upset by this contact, which could alter the mixture’s general characteristics. The solubility or behavior of the polymer may change in the presence of chloride ions, which could modify the texture or viscosity of the combination. That explains the texture change from a semi-solid to a more solid, snow-like substance.
Cornstarch: Cornstarch is classified as a nonpolar molecule. Due to their nonpolar nature, these compounds have very weak intermolecular forces – such as van der Waals interactions. In the case of sodium polymethacrylate – a polar polymer containing carbonyl groups – the nonpolar nature of corn starch particles may not lead to strong interactions with the polymer chains. As a result, the addition of baby powder might have minimal effects on the overall consistency or texture of the mixture.
The properties of sodium polymethacrylate are ideally suited for its use in diapers and feminine hygiene products. Suggest at least 3 other uses for this polymer.
Agriculture: Sodium polymethacrylate can be employed as a soil conditioner in agriculture. Its water-absorbing capacity allows it to retain moisture in the soil, providing a more favorable environment for plant growth. This polymer helps prevent water loss from irrigation and enhances water efficiency in arid regions. It also assists in reducing the frequency of watering, which can lead to water conservation and improved crop yields.
Packaging and Transportation: Sodium polymethacrylate can be used in packaging materials for products sensitive to moisture or prone to damage due to water exposure. By incorporating this polymer into packaging films or desiccant sachets, it can help maintain the dryness of the packaged goods and protect them from moisture-related deterioration. This application is particularly valuable for electronics, pharmaceuticals, and certain food products.
Water-blocking Cables and Pipes: Sodium polymethacrylate can be used in cables and pipes to prevent water ingress or leakage. Incorporating the polymer into cable jackets or pipe coatings can act as a barrier that swells upon contact with water, effectively sealing any potential entry points. This application is particularly useful in telecommunications, underground utility systems, and infrastructure projects where protection against moisture is crucial to maintain optimal performance and prevent damage.
Show the condensation reaction that results when the monomer(s) reacts to form polymethacrylate.
Polymethacrylate refers to a polymer formed by the condensation polymerization of methacrylic acid. The condensation reaction typically involves the loss of a small molecule such as water or methanol.
The condensation reaction for the formation of polymethacrylate, using methacrylic acid as the monomer, can be represented as follows:
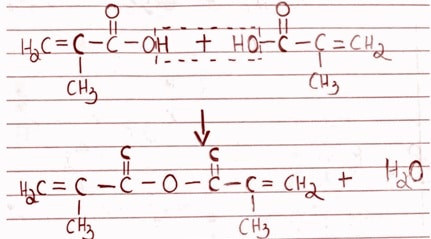
In this reaction, two molecules of methacrylic acid (H2C=C(CH3)CO2H) combine to form a dimer, where the two monomers are linked by an ester bond. The release of one molecule of water (H2O) occurs as a byproduct of this condensation reaction.
It is important to note, however that polymethacrylate is formed through addition polymerization. The monomer used in the addition polymerization is known as methyl methacrylate and during the reaction, the monomers add together without eliminating any small molecules as byproducts. The reaction typically involves the activation of the double bond present in the monomer, leading to the formation of a chain of repeating units.
Polymers are often thought to be manufactured in immense chemical plants. Although many are, there is a large group of polymers which are natural (biopolymers). Describe several natural polymers and how they are utilized in our society or in our bodies. Pick one and show its reaction.
- Cellulose: Cellulose is a biopolymer found in the cell walls of plants. It is the most abundant natural polymer on Earth and has various applications. Cellulose fibers are used to produce textiles, paper, and cardboard. It is also used as a thickener in food products and as an additive in pharmaceuticals. In our bodies, cellulose is an important dietary fiber that aids in digestion and promotes bowel regularity.
- Chitin: Chitin is a biopolymer found in the exoskeletons of arthropods (such as insects and crustaceans) and the cell walls of fungi. It is a tough and flexible polymer with various applications. Chitin is used in the production of biodegradable films, coatings, and packaging materials. It is also utilized in the medical field for wound dressings, surgical sutures, and drug delivery systems.
- Proteins: Proteins are biopolymers composed of amino acid chains and are essential components of our bodies. They have diverse functions, such as acting as enzymes, hormones, antibodies, and structural components. Proteins are crucial for muscle development, immune system function, and overall health. In industry, proteins are used in food production, pharmaceuticals, cosmetics, and biotechnology.
Chitin deacetylation reaction:
Chitin can undergo a deacetylation reaction to produce another natural polymer called chitosan. The reaction involves the removal of acetyl groups from chitin, resulting in the conversion of N-acetylglucosamine units into glucosamine units. This reaction can be achieved by treating chitin with an alkaline solution, typically using sodium hydroxide (NaOH).
The reaction can be represented as follows:
(N-acetylglucosamine) n + NaOH → (Glucosamine) n + CH3COONa
In this reaction, n represents the number of repeating units in the chitin polymer chain. The deacetylation reaction leads to the formation of chitosan, which possesses different properties compared to chitin. Chitosan has increased solubility and improved biocompatibility, making it useful in various applications, such as wound healing, drug delivery systems, and antimicrobial coatings.
- The structure given below is a polymer classified as a polyamide. This polymer is stable in temperatures above 350°C, and can be used as a protective coating on hot surfaces. What monomers were used to make this polyamide? What polymerization reaction likely took place to create it? Show this reaction.
The monomers of the polyamide include Diamino compounds and polycarboxylic acid molecules. The monomers of the mentioned polymers are as follows:
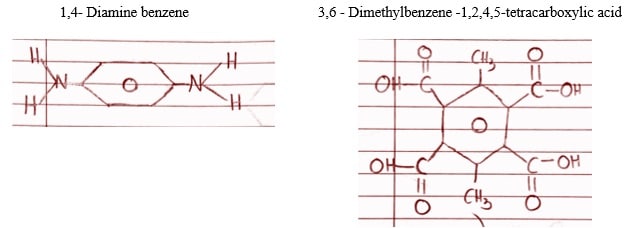
The condensation polymerization reaction can be represented as:

This reaction used to create this polyamide is known as a condensation reaction, which involves joining monomers with the elimination of a small molecule, typically water, as a byproduct. In this reaction, the hydrogen present in the amino group monomer reacts with the carboxylic group in the other monomer, releasing water and the formation of amide linkage between nitrogen and carbon. The formation of this amide group results in a polymer chain known as polyamides. There are various types of polyamides and some common examples include nylon, wool etc.


