Research Question
How does salinity affect the seed germination of Solanum lycopersicum?
Introduction
Relevance to Real world
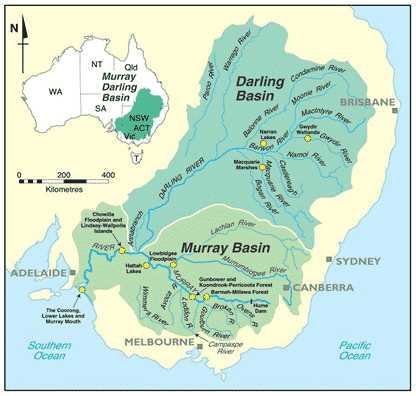
Covering 14% of Australia and home to over two million people, the Murray Darling Basin is Australia’s largest and most iconic river system (figure 1) (McCormick, 2015). As over one-third of Australia’s food supply is produced by the Basin (Discover Murray, 2016), increased salinity levels affect both Australia’s agriculture and the overall economy.
Deep beneath the Murray Darling Basin, lies salty groundwater which flows through the river, out Murray’s mouth, and into the ocean (Government of South Australia, 2019). Before the development of agriculture, local farming regions were covered by native vegetation. The native vegetation thrived from the rainfall, absorbing the water for survival. Therefore, less water is soaked up by the soil and moved into the salty groundwater.
Since there has been an increase in demand for agricultural supplies, the native vegetation has been cleared; replaced with irrigated crops. Resultantly, more water is absorbed by the soil, increasing the amount of water moving into the salty groundwater, transferring into the river. Key species including the Eucalyptus camaldulensis (river redgums) have specific salinity thresholds that are notwithstanding these rising levels; causing habitat loss for many species (Government of South Australia, 2019).
Furthermore, loss of habitat creates a struggle for survival of both flora and fauna as they are more vulnerable to environmental pressures.
Process of seed germination
The initial phase of plant growth is seed germination. Seed germination is recognised as the original sprouting from the seeds to the development into plants (Robb, 2017). Before germination occurs, environmental conditions must trigger the conversion of ‘food’ being released from the seed endosperm to energy for the seed embryo; conditions include water, temperature, sunlight and air.
Relative oxygen species (ROS) are chemically reactive species that contain oxygen (such as peroxides). When ROS are released, O2 oxidises into different substances creating oxidative stress. ROS play a large role during seed germination (Arbor Assays, 2017). Maintaining the correct ROS levels between a small range will ensure germination of the seeds.
Varying to low, seeds will never leave dormancy and varying too high and the seeds will suffer extreme oxidative damage during seed and will be non-viable. During the early stages, seedlings are most vulnerable and heavily rely on water intake. Once a seed is exposed to favourable conditions, water and oxygen are absorbed through the seed coat.
The embryo cell begins to swell and the radicle emerges from the seed coats (figure 2) (University of Illinois Extension, 2019).

Effect of Salinity on Seed Germination
Plant stress is any unfavourable factors that alter a plant’s equilibrium, growth or development (Lichtenthaler, 1996). During the development of seeds, the breakdown of ROS creates oxidative stress. In the presence and in increasing concentrations, salinity triggers a high accumulation of ROS levels, causing damage (AbdElgawad, et al., 2016). Dormancy is the prolonged development of seed embryos due to unsuitable conditions (R. B. & S. B., 1977) and until the appropriate environmental conditions are met, seeds will not germinate.
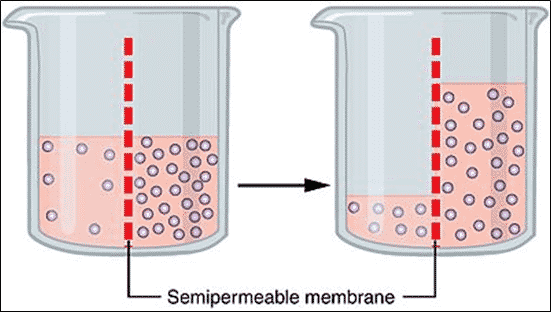
As mentioned above, water intake is vital to the development of seeds and exposure to salt stress increase the requirement of water. Osmosis is the diffusion of water from an area of high concentration water molecules to another in a lower concentration through a partially permeable membrane (BBC Bitesize, 2019).
In plants, water is attracted to the region of the cell membrane with the highest concentration of salt. If the highest concentration is outside the cell membrane, water escapes the cell to bond with the sodium chloride (figure 3) (Crowe, 2019). Osmotic stress is the abrupt change in the solute concentration around a cell, creating a rapid change in the movement of water across its cell membrane.
In the presence of increased salt concentrations, osmotic stress occurs as the water moves across the membrane towards the salt. Furthermore, increased sodium chloride concentrations accumulates the soluble solutes around the seeds, increasing the osmotic pressure. Additionally causing excessive uptake of ions, resulting in toxicity in the plant (Jones, 1986).
Applications to Similar Experiments
Similar studies, where Solanum Lycopersicum seeds were treated with increasing concentrations of sodium chloride, have shown that the germination rate decreased with increasing salinity and the germination period took longer (Singer, 1994; El-Habbasha et al., 1996; Cuartero and Fernández-Muñoz, 1999). Correlating with these studies are the works of Meza et al. (2007), observing the same trend: germination decreased with increased salt concentrations.
While the following investigation will produce different values for the percentage of seeds germinating, based on the theory, the central relationship shared between the independent and dependent variables will be inverse. Different plants have different salinity thresholds. For this experiment, it is important to consider plants that are salt-tolerant (halophytes) to enable real-world applications.
Solanum Lycopersicum (tomato seeds) have an average tolerance. Therefore, they are suitable for determining how salinity affects seed germination.
Aim
To determine the effect of increasing concentrations of sodium chloride on the seed germination of Solanum Lycopersicum (tomato plant).
Hypothesis
It is predicted that if the concentration of sodium chloride is increased, then the percentage of seed germination will decrease.
Hypothesis Justification
Before seeds can germinate, certain conditions must be suitable for the seed embryo to survive; temperature, water, sunlight and air (White, 2016). During dormancy, tomato seeds store ‘food’ in their endosperms. Adequate amounts of the correct water quality, and the ‘food’ will release from the endosperm and convert into energy for the development of the embryo.
During this experiment, the increasing salinity concentrations should create less acceptable water quality for the ‘food’ to be released and slowing the development of the embryo.
The hypothesis is validated by a study by Miquel et al., who during a similar experiment documented that salinity can affect the germination of seeds by producing osmotic potential, preventing water uptake (Kaymakanova, 2014). In this study, the results showed a decrease in the percentage of seeds that germinated as the salinity concentration increased.
Experimental Design
Table 1. Experimental design
| Variable Type: | Variable: | Description: | How it was measured/ controlled: |
| Independent | Concentration of Sodium Chloride | Concentration of Salt for trials (g): 0% (normal water)2%4%6% | Measured using a milligram scale based on measurements of parts per million/milligrams (mg) before the experiment began. |
| Dependent | Percentage of seed germination | Percentage of seed germination is the percentage of seeds that clearly show the emerged radicle. | The percentage of germination is measured by the number of radicles that have emerged within that petri dish. |
| Controlled | Amount of sprays for the seeds each day | The cotton wool buds lining the petri dishes must be kept damp at all times. | To ensure the cotton wool is always damp, each day the experimentalist will lightly spray each petri dish 6 times, targeting all areas. To ensure that the seeds do not drown, each time, the petri dish is investigated for loose drops of water. |
| Time of Experimentation | For one week, the experiment occurs twice each day. The seeds are sprayed at 7am and 5 pm. | Every day an alarm is kept to remind the experimentalist when to spray the trials, therefore keeping consistency. | |
| Source of water | The source and quality of water is controlled so that the trials will equally show the results. | The concentrations of salt are changed for the experiment to be 0%, 2%, 4% and 6%. To make these concentrations, see the calculations in appendix 2. | |
| Time duration for experiments | During the experiments, it takes the same amount of time every day. | Spray bottles allow the whole surface area of the cotton wools to be covered at equal amounts. If I was to drop a small amount of water into the petri dishes, the water wouldn’t reach some of the seeds. | |
| Spray bottles, petri dishes, cotton wool and the type of tomato seed | The spray bottles, petri dishes, cotton wool and the types of tomato seed were all bought from the same supplier. | To ensure reliability, both the spray bottles and tomato seeds were all bought from Bunnings and then the petri dishes and the cotton wool were supplied by the school. | |
| Uncontrolled | Oxygen supply | Availability of oxygen in the air. | The oxygen supply is uncontrollable as the experiment is placed inside on a cupboard. |
| Ambient Temperature | The temperature of the room throughout the trials. | The ambient temperature does vary, however, there is nothing that can be changed to keep the temperature as a constant. |
Materials
- 1 x Scales
- 2L x tap water
- 60g x Sodium Chloride
- 4 x 500ml Spray bottles
- 1 x Thistle Funnel
- 1 x Stirring rod
- 20 x Petri dishes
- 140 x Cotton wool
- 1 x Spoon
- 1 x Table
- 1 x Permanent marker
- 400 x Tomato seeds
Method
Procedures
- Label
20 clear plastic petri dishes as the following, with the correct starting date:
- Table 2 – Example labels for the clear plastic petri dishes.
| Trial 1 – 0% [date] | Trial 1 – 2% [date] | Trial 1 – 4% [date] | Trial 1 – 6% [date] |
| Trial 2 – 0% [date] | Trial 2 – 2% [date] | Trial 2 – 4% [date] | Trial 2 – 6% [date] |
| Trial 3 – 0% [date] | Trial 3 – 2% [date] | Trial 3 – 4% [date] | Trial 3 – 6% [date] |
| Trial 4 – 0% [date] | Trial 4 – 2% [date] | Trial 4 – 4% [date] | Trial 4 – 6% [date] |
| Trial 5 – 0% [date] | Trial 5 – 2% [date] | Trial 5 – 4% [date] | Trial 5 – 6% [date] |
- Label
the spray bottles accordingly:
- Table 3 – Example labels for the spray bottles.
| 0% Concentration [date] | 2% Concentration [date] | 4% Concentration [date] | 6% Concentration [date] |
- Calculate the correct amount of salt needed for the concentrations (appendix 1).
- With the scales, weigh the correct amount of salt needed for the spray bottles (10g, 20g and 30g) and place into the bottom of the respective empty spray bottles using the funnel.
- Pour
500mLs of tap water into each bottle.
- Using the stirring rod, combine the salt and tap water for the spray bottles: 2%, 4% and 6%. Stop stirring once the salt has completely dissolved.
- Line
the petri dish with 8 cotton wool balls. For full coverage of the dish, stretch
the cotton wool so there are no gaps showing (figure 4).
- Repeat step 6 for the following 19 trials.
- Count 20 seeds for the petri dish and gently place on top of the cotton wool (figure 5).
- Seeds need to be placed at an approximate even distance away from each other.
- Repeat step 7 for the following 19 trials.
- Spray the seeds in the petri dish with the respective spray bottle until it is completely wet (ten sprays).
- There should be no standing water at the bottom of the dish.
- Repeat step 8 for the following 19 trials.
- Place the dish in the examination location (on a table at ambient temperature).
- Repeat step 9 for the following 19 trials.
- Twice a day, spray the correct dishes with the respective spray bottle (ten sprays).
- Before respraying the trials each day, record any observations.
- Repeat step 9 and 10 for the following 19 trials.
- After seven days of trialing, count and record the number of seeds which have germinated in each dish.


Preliminary Results
Table 4 – Number of Seeds Germinating at increasing sodium chloride concentrations
| Sodium Chloride Concentration (%) | |||||
| 0 | 2 | 4 | 6 | ||
| Number of Seeds Germinated | 1 | 12 | 5 | 0 | 0 |
| 2 | 15 | 3 | 0 | 0 | |
| 3 | 13 | 4 | 0 | 0 | |
| 4 | 11 | 0 | 0 | 0 | |
| 5 | 14 | 2 | 0 | 0 |
Table 5 – Percentage of Seeds Germinating at increasing sodium chloride concentrations (calculation method is shown in Appendix 1)
| Sodium Chloride Concentration (%) | |||||
| 0 | 2 | 4 | 6 | ||
| Percentage of Seeds Germinated | 1 | 60 | 25 | 0 | 0 |
| 2 | 75 | 15 | 0 | 0 | |
| 3 | 65 | 2 | 0 | 0 | |
| 4 | 55 | 0 | 0 | 0 | |
| 5 | 70 | 10 | 0 | 0 |
Graph 1 – Preliminary Results of Seed Germination

The concentrations of salinity tested for this experiment is 0%, 2%, 4% and 6%. As predicted, the most percentage of germination is from the concentration with no salinity. However, after 2%, there is no germination from the seeds. Solanum lycopersicum is asalt-tolerant plant, therefore there should be an increase in the germination percentages.
In comparison to an investigation with similar purpose (Demir, Mavi, Ozcoban, & Okcu, 2003), the results showed that 4% and 6% still had small percentages of seeds germinating. Therefore, the method needs to be altered to increase the seeds germinating. Observations within the preliminary trials revealed that seeds were becoming dehydrated as there was not enough water supplied, the cotton wool the seeds were placed on was thinly spread and the location was in a windy environment.
Improving these factors could potentially show the desired results.
Table 6 – Method Refinements
| Method Refinements | Justification |
| Location | In the preliminary trials, the petri dishes were placed on a table outside, exposed to light, wind and varying temperatures. As the location is a controlled variable, I decided it was best if I placed them on a table inside a bedroom, at an ambient temperature. |
| Number of sprays | Observations from the preliminary trials include dryness of the cotton wool and crystallisation of the sodium chloride. Both factors are due to low amounts of water. Therefore, by increasing the number of sprays from six to ten, the trials are always damp. |
| Amount of cotton wool | Cotton wool, during the experiment, acts as soil in the real world context. Cotton wool absorbs the water, but maintains dampness for long periods of time, depending on the amount of sprays and the amount of cotton wool. Therefore, the more cotton wool there is, the longer the seeds will be in damp conditions. Therefore, the amount of cotton wool used has been increased from five pieces to eight. |
Safety Considerations / Risk Assessment
+ A risk assignment has been attached, see appendix 2.
Table 7 –The table below exhibits the safety considerations for this experiment.
| Object | Hazards | Control Measures |
| Tomato Seeds | Allergies to seeds. Avoid eating the seeds as they may be treated with toxic fungicides. | Keep dry |
| Water | Spillage. Chemical contamination | If water is spilled, clean immediately or use a wet floor sign to indicate the spillage. Water in a laboratory should not be drunk, due to the possibility of chemical contamination. |
| Spray Bottles | May be used to spray others. | Use responsibly. Avoid the eyes |
Results
Raw Data
Table 8 – Number of Seeds Germinating at increasing sodium chloride concentrations
| Sodium Chloride concentration (%) | |||||
| 0 | 2 | 4 | 6 | ||
| Trials | 1 | 17 | 12 | 8 | 3 |
| 2 | 15 | 13 | 11 | 5 | |
| 3 | 18 | 10 | 10 | 4 | |
| 4 | 19 | 9 | 9 | 3 | |
| 5 | 16 | 15 | 6 | 2 |
Processed Data
+ The following results tables has been calculated using the method in (appendix 1)
Table 9 – Percentage of Seeds Germinating at increasing sodium chloride concentrations
| Sodium Chloride concentration (%) | |||||
| 0 | 2 | 4 | 6 | ||
| Trials | Trial 1 | 85 | 60 | 40 | 15 |
| Trial 2 | 75 | 65 | 55 | 25 | |
| Trial 3 | 90 | 50 | 50 | 20 | |
| Trial 4 | 95 | 45 | 45 | 15 | |
| Trial 5 | 80 | 75 | 30 | 10 |
Table 10 – Number of Seeds Germinating at increasing sodium chloride concentrations
| Sodium Chloride concentration (%) | ||||
| 0 | 2 | 4 | 6 | |
| Average Percentage of seeds germinated | 85 | 59 | 44 | 17 |
Graph 2 – Percentage of seeds germinated
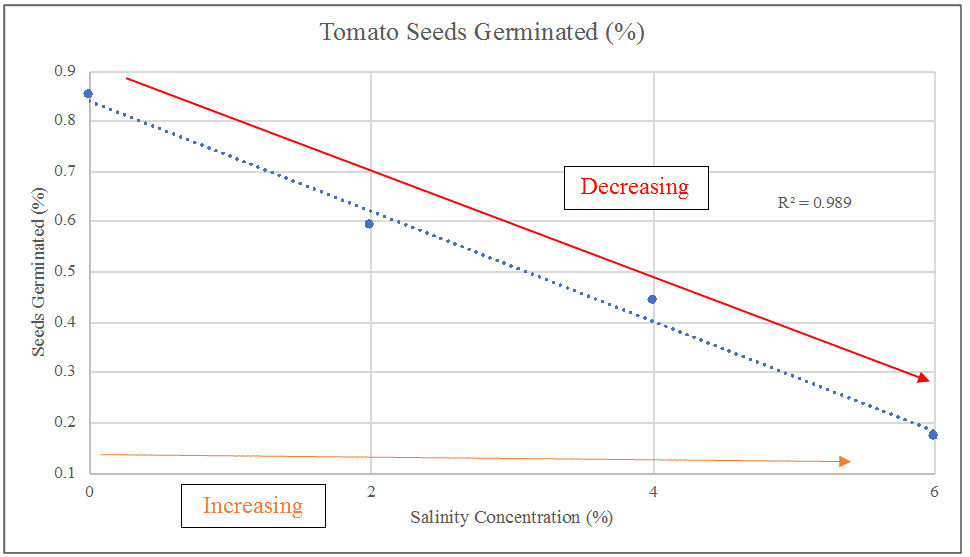
Graph 2 reveals an inverse relationship between salinity and seed germination: as salinity increases, seed germination decreases. These results display a linear decrease and is proved to be considerably accurate as the R2 value is 0.989.
Observations
- Over the course of the trialing period, most of the seeds began to germinate after the fourth day.
- During preliminary trials, with only 6 sprays, the cotton wool became hard and crystallised. However with 10 sprays, the cotton wool maintained the perfect dampness with no crystallization. Salt did develop around the seeds.
- Observing the seeds over the trialing period, showed that as the salinity increased, the less seeds were germinating.
Discussion
Analysis
Before seeds can germinate, the surrounding environment must display favourable conditions, causing the release of the energy stored within the embryo in the form of food. Seeds rely on the water intake to receive the proper nutrients to survive. In Graph 2, the control trials (no sodium chloride) reached an average 85% seeds germinated. As the percentage of salinity increased, there is a noticeable decrease within the trials: 59%, 44% and finally 17% respectively.
The accuracy of these results are supported by the R2 value which is 0.989. The closer the value is to 1, the more accuracy and close fitting to a trend line the data is. Thus it is fair to say that overall, the trials show a linear decrease. These results correlate with several different studies, supporting the hypothesis that there is a clear inverse relationship shared between both the independent and dependent variable (the concentration of sodium chloride and the percentage of seed germination respectively).
For the survival of any seed during the early stages, naturally there is a small percentage of sodium chloride. While a key component of the process of seed germination is relative oxygen species (ROS), when exposed to the salinity, oxygen species react to form oxidative stress on the seed (Arbor Assays, 2017). The levels of ROS are very delicate, too low leads to dormancy and too high causes damage and therefore it is important to understand the variables that can alter the equilibrium.
In relation to this investigation, approximately none of the seeds reached ROS levels too low to remain in dormancy, but rather they were too high the trials became damaged. Larger concentrations of salinity create an accumulation of high ROS levels, thus it is reasonable to say the seeds were damaged because of the intensity of the salinity (AbdElgawad, et al., 2016). Concentrations 0-2% showed the highest and most successful percentage of seed germination, in comparison to 4-6% which showed the lowest.
There are only a few requirements for the germination of seeds: proper oxygen, temperature, water, and nutrients. If seeds do not receive these vital components, they will experience dormancy. These components trigger the process of ‘food’ being released from the endosperm into energy for the seed embryo. Water and oxygen are absorbed through the seed coat (figure 2) and the radicle will begin to emerge. In this investigation, different concentration of salinity was tested on the seeds.
Water within the plant cell is attracted to the salt ions, and will quickly move across the cell membrane to be with the highest concentration of sodium chloride. This process is known as osmosis. However, under increasing sodium chloride concentrations, osmosis begins to be another form of stress on the plant. During the experiment, it is appropriate to conclude that within the larger sodium chloride concentrations, osmotic stress was interfering with the germination. As a result of the relationship between the variables, the larger the concentration, the less percentage of seed germination.
It can be expected that more water is needed to fully hydrate the seeds to maintain the ROS levels (Zhang, Irving, Tian, & Zhoua, 2012). Consequently, the more water required increases the osmotic pressure. Furthermore, increased sodium chloride concentrations accumulate the soluble solutes around the seeds, increasing the osmotic pressure (Jones, 1986).
Due to the osmotic pressure created by sodium chloride, water can no longer enter the seed coat; instead, salt enters and causes toxicity to plants. This leads to underdeveloped growth and reduction in yield (Cocoponics, 2011). Based on the observations, the seeds with the higher concentrations all appeared dehydrated, in comparison to the 0-2% trials which were damp. The dehydration and resultantly little germination of the seeds is supported by the concept of osmotic pressure.
Overall within the experiment, the comparison percentage germination between 0% concentration and 6% concentration is 68%. The results show that the best concentration for the most percentage of seed germination occurs under the concentration of no salinity (0%). However, in relation to the Murray Darling Basin, there will always be some form of salinity flowing through the basin to escape at the Murry mouth, due to the salty groundwater below the surface.
While the ocean has a salinity percentage of 3.5% (Science Daily, 2017), the Murray Darling Basin is considered a combination of many fresh water rivers. Therefore, the average salinity percentage must be lower than 3.5%. Concentrations of 4% and 6% would have devastating effects on the ecosystem where as 2% will not. From the above results, it can be concluded that if salinity percentage of the Murray Darling Basin remains below 2%, the ecosystem will flourish.
Concluding the results from several different reports and studies (Singer, 1994; El-Habbasha et al., 1996; Cuartero and Fernández-Muñoz, 1999) and the results from this experiment, the hypothesis has been supported, outlining the clear linear inverse relationship between the concentration of salinity and the percentage of seed germination.
Evaluation
During this investigation, the experiment produced the results that were hypothesised. However, the results would not have been as valid if preliminary trials were not completed. Preliminary trials aim to insure accuracy and effectiveness within the method. In comparison to the primary data, the preliminary trials did not perform as well, due to not enough water, in a dry sunny location and too many cotton wool balls.
Resultantly, these issues were fixed in the method refinements. Despite the actions to increase reliability and accuracy, there are a few sources that can risk and effect the overall results due to errors. The table below (table 11) outlines the sources of potential error.
Table 11 – Sources, effect and solution for potential errors
| Source of Error | Error | Effect |
| Number of Trials | Only 5 trials were conducted for each concentration. As five is a small number, the results are very limited and provide a large amount of error. | Having a smaller number of trials limits the overall results and therefore the conclusion of the experiment (whether the hypothesis is supported or rejected). A smaller number of trials decrease the reliability of this experiment. |
| Number of Concentrations and the percentage of each | During the experiment the concentrations used were 0%, 2%, 4% and 6%. These concentrations could potentially limit the results. | During the experiment, the results found that even at 6% salinity, there were a few seeds still germinating. This limits the results as there could have been germination at 8%. |
| Trialing period | Trialing period for the seed germination only ran for seven days. The cut off for trials could limit the results. | The trialing period ran for only seven days, despite there being enough time during the investigation to increase the number of days. Overall, different percentages or trends may have emerged and therefore effecting whether the hypothesis is supported or rejected. |
| Type of plant species | The experiment was only limited to Solanum lycopersicum (tomato seeds). | Solanum lycopersicum (tomato seeds) are considered salt-tolerant plants. When determining the effect of salinity on germination, both halophytes and glycophytes. Therefore, the results only provide the answer to how do salt-tolerant plants react to salinity, instead of including all types. |
| Not washing the seeds before | None of the seeds were washed before the experiment occurred. | An observation on the germination of the seeds was that it was only within the last few days did the seeds begin to germinate. A potential error is not washing the seeds before the experiment. This alters the seeds natural defences against environmental stressors, ultimately allowing an increase in the rate of germination (Rhoades, 2018). |
| Parallax Error | Displacement of measurement positions can occur when viewing measuring equipment at an oblique angle. Parallax error can cause altered perceptions and reading measurements. | Contributes to minor errors within measuring amount of materials for different concentrations and measuring the radius of inhibition. This further affects accuracy of results. |
Improvements to experiment
For better and improved results for future experiments, improvements can be used to increase the accuracy. An adjustment as simple as increasing the number of trials from 5 per concentration to 10 per concentration creates a more reliable data plot as the results are focused on a widespread of data. More trials allows room for outliers and can fully represent the data being displayed.
The more trials completed, allows for the experimentalist to give a properly educated conclusion after the experiment. As this investigation is specifically focused on the vegetation surrounding the Murray Darling Basin, future experiments should expand the types of seeds being studied to the rather prominent native halophyte seeds that are crucial to the environment.
Furthermore, increasing the range between the concentrations, and the different types of seeds, allows for experimentalists to determine which seeds have a higher/ lower salt tolerance and will survive in the changing environment. Additionally, this will create a widespread of data, allowing for all trends and outliers.
Another improvement to further this study can be to extend the trialing period focus on growth rates as well as germination. If future experiments can determine the growth rates of surrounding local plants to the Murray Darling Basin and the seed germination while exposed to environmental stressors, farmers can determine better seeds to plant in certain paddocks based on those factors; increasing both the agricultural and economical value of Australia.
Conclusion
This investigation supported the hypothesis that if the concentration of sodium chloride is increased, then the percentage of seed germination will decrease. After the first few days, the seeds began to germinate; the most being 0% followed in a linear decrease by 2%, 4% and finally 6%.
The importance of these results is that it can give an idea as to the appropriate levels of salinity the Murray Darling Basin must be maintained at in order to achieve a thriving ecosystem. In the future, further studies can extend to the growth rates of plants and environmental factors cause harm. Further studies can be used to support the decisions made affecting the Murray Darling Basin to minimise the impact on the environment.
Appendices
Appendix 1 – Calculations

References
AbdElgawad, H., Zinta, G., Hegab, M. M., Pandey, R., Asard, H., & Abuelsoud, W. (2016, March 8). High Salinity Induces Different Oxidative Stress and Antioxidant Responses in Maize Seedlings Organs. Retrieved from US National Library of Medicine – National Institutes of Health: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4781871/
Arbor Assays. (2017, October 5). Effects of Oxidative Stress on Germination. Retrieved from Arbor Assays: http://www.arborassays.com/effects-oxidative-stress-germination/
BBC Bitesize. (2019). Transport in cells – AQA. Retrieved July 17, 2019, from BBC Bitesize: https://www.bbc.com/bitesize/guides/zs63tv4/revision/4
Cocoponics. (2011, October 3). How salt affects Seed Germination. Retrieved from Cocoponics Community: http://www.cocoponics.co/germination-2/salt-affects-seed-germination
Crowe, B. (2019). What Does a High Concentration of Salt Do to a Cell Membrane? Retrieved July 17, 2019, from Seattle PI: https://education.seattlepi.com/high-concentration-salt-cell-membrane-4414.html
Cuartero, J., & Fernández-Muñoz, R. (1998, November 30). Tomato and salinity. Scientia Horticulturae, 78(1-4), 83-125.
Demir, I., Mavi, K., Ozcoban, M., & Okcu, G. (2003). Effect of salt stress on germination and seedling growth in serially harvested aubergine (Solanum melongena L.) seeds during development. Israel Journal of Plant Sciences, 51(2), 125-131.
Discover Murray. (2016). Murray Darling Basin. Retrieved from Discover Murray: http://www.murrayriver.com.au/about-the-murray/murray-darling-basin/
El-Habbasha, K., Shaheen, A., & Rizk, F. (1998). Germination of some tomato cultivars as affected by salinity stress condition. Egyptian Documentation and Information Centre for Agriculture, 23(2), 179-190.
Government of South Australia. (2019, March 3). Salinity. Retrieved from Natural Resources SA Murray Darling Basin: https://www.naturalresources.sa.gov.au/samurraydarlingbasin/water/river-murray/issues-for-river-health/salinity
Jones, R. (1986, June). High salt tolerance potential in Lycopersicon species during germination. Euphytica, 35(2), 575–582. Retrieved from Springer Link: https://link.springer.com/article/10.1007/BF00021866
Kaymakanova, M. (2014). Effect of Salinity on Germination and Seed Physiology in Bean (Phaseolus Vulgaris L.). Agricultural University Plovdiv. Bulgaria: Taylor and Francis Group, LLC.
Kranner, I., Minibayeva, F. V., Beckett, R. P., & Seal, C. E. (2010, September 10). What is stress? Concepts, definitions and applications in seed science. Retrieved from New Phytologist: https://nph.onlinelibrary.wiley.com/doi/10.1111/j.1469-8137.2010.03461.x
Lichtenthaler. (1996). Vegetation stress: An introduction to the stress concept in plants. PLANT PHYSIOLOGY, 4-14. Retrieved from Web of Science: http://cel.webofknowledge.com/InboundService.do?customersID=atyponcel&smartRedirect=yes&mode=FullRecord&IsProductCode=Yes&product=CEL&Init=Yes&Func=Frame&action=retrieve&SrcApp=literatum&SrcAuth=atyponcel&SID=C3UyoChxpOizTuv7f5a&UT=WOS%3AA1996UL69800002
Mavi, K. (2008, October). Effect of salt and osmotic stresses on the germination of pepper seeds of different maturation stages. Retrieved from SciELO: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-89132008000500004
McCormick, B. (2015). Parliament of Australia. Retrieved July 16, 2019, from Murray-Darling Basin management: https://www.aph.gov.au/About_Parliament/Parliamentary_Departments/Parliamentary_Library/pubs/BriefingBook44p/MurryDarlingBasin
Meza, N., Arizaleta, M., & Bautista, D. (2007). Efecto de la salinidad en la germinación y emergencia de semillas de parchita. Passiflora edulis f. flavicarpa, 24(1), 69-80.
R. B., T., & S. B., H. (1977, June). Annual Reviews. Retrieved July 16, 2019, from Dormancy in Seeds: https://www.annualreviews.org/doi/pdf/10.1146/annurev.pp.28.060177.001555
Rhoades, H. (2018, August 5). How To Soak Seeds Before Planting And The Reasons For Soaking Seeds. Retrieved from Gardening How To: https://www.gardeningknowhow.com/garden-how-to/propagation/seeds/soaking-seeds.htm
Robb, A. (2017). Study.com. Retrieved July 16, 2019, from What is Seed Germination? – Definition, Process, Steps & Factors: https://study.com/academy/lesson/what-is-seed-germination-definition-process-steps-factors.html
Science Daily. (2017, August). Sea water. Retrieved from Science Daily: https://www.sciencedaily.com/terms/seawater.htm
Singer, S. M. (1997). Germination responses of some tomato genotypes as affected by salinity and temperature stress. Egyptian Documentation and Information Centre for Agriculture, 21(1), 47-64.
University of Illinois Extension. (2019). Background Information. Retrieved from The Great Plant Escape: https://web.extension.illinois.edu/gpe/tg/c3-background.html
White, N. (2016, May 6). Conditions Necessary for Germination. Retrieved July 17, 2019, from Tisiawikispace2016: https://sites.google.com/site/tisiawikispace2016/mysciencewikipage/conditionsnecessaryforgermination
Zhang, H., Irving, L., Tian, Y., & Zhoua, D. (2012). Influence of salinity and temperature on seed germination rate and the hydrotime model parameters for the halophyte, Chloris virgata, and the glycophyte, Digitaria sanguinalis. South African Journal of Botany, 78, 203-210.

Good Facts