Stearic Acid: Production, Use and Economical Impact
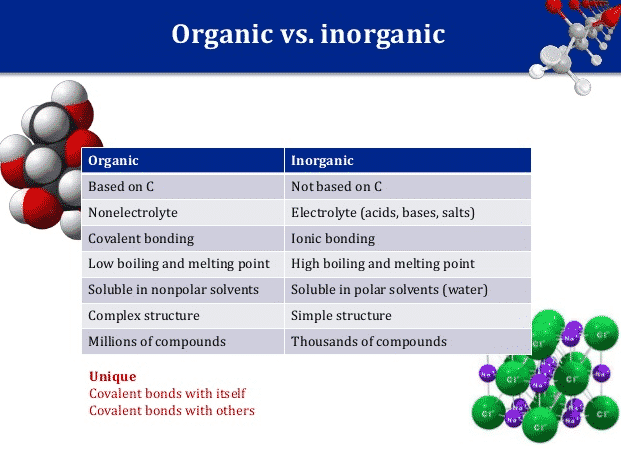
Stearic acid is known as Octadecanoic acid, one of the common long-chain fatty acids being a very useful saturated fatty acid present as a combined form and a major component in products such as cocoa and shea butter, and various plant and animal fats. Having 18-carbon as its backbone, the chemical formula of stearic acid…