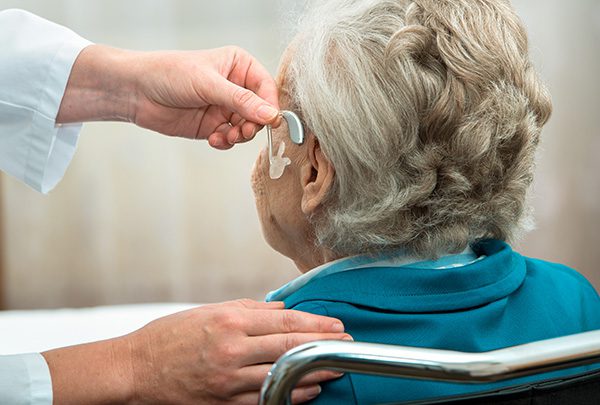
Effects Associated with Hearing Loss Among the Elderly
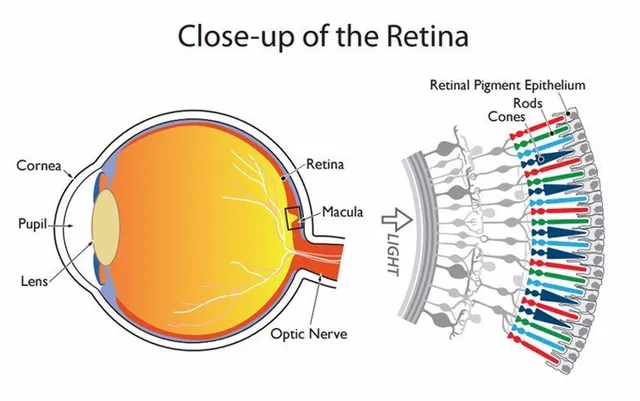
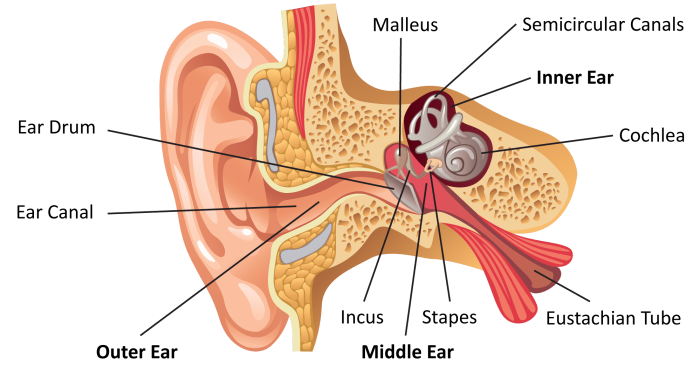
Humans lose hair, sight, teeth, and muscles with age. Hearing is not any different. There are many reasons people lose their hearing ability as they age. Normal wear and tear are one of the reasons people’s hearing abilities decrease with age, damaging, breaking, and wearing out of hair cells. Sometimes the cells just die. The…