William completed his Bachelor of Science and Master of Arts in 2013. He current serves as a lecturer, tutor and freelance writer. In his spare time, he enjoys reading, walking his dog and parasailing.
Article last reviewed: 2022 | St. Rosemary Institution © 2010-2025 | Creative Commons 4.0
Cardinal Joseph Bernardin was born on April 2,1928, in Columbia, South Carolina. He was son of Mrs. Marie M. Simon Bernardin, and the late Joseph Bernardin. Cardinal Bernardin attended Catholic and public schools and the University of South Carolina in Columbia. He was later accepted as a candidate for the priesthood by Most Rev. John…
Nicholas Ferrar was assumed to be born in 1592. I have found that his most probable birth date was in February of 1593. This is due to the usual calendar confusion: England was not at that time using the new calendar adopted in October 1582. It was 1593 according to our modern calendar, but at…
Bishop Francis X. Ford was a well educated, enormously gentle man that was kicked, beaten, insulted, and surrounded by hatred. All this because of one man’s beliefs. He was born in Brooklyn in 1892. He was the founder of the Maryknoll Missionaries and was the first bishop of Kwantung, China. He was killed in the…
Barnabas was a native of the island of Cyprus. His birthplace makes him a Jew of the Diaspora, the dispersion of Jews outside Palestine or modern Israel. He was originally named Joseph but the apostles called him Barnabas, he probably acquired this name because of his ability as a preacher. The name Barnabas was understood…
The Bhagavad-Gita begins with the preparation of battle between the two opposing sides: on the left stands the collected armies of the one hundred sons of Dhritarashtra and on the right lie the soldiers of the Pandava brothers. Warring relatives feuding over the right to govern the land of Kurukshetra, both forces stand poised and…
A heresy which arose in the fourth century, and denied the Divinity of Jesus Christ. DOCTRINE First among the doctrinal disputes which troubled Christians after Constantine had recognized the Church in A.D. 313, and the parent of many more during some three centuries, Arianism occupies a large place in ecclesiastical history. It is not a…
The Unity of the Unknown and the Eternal Security: The Anglo-Saxon Belief in Christianity and Fate Imagine a life in which one is simply a pawn at the hands of a mysterious higher force stumbling and meandering through life’s tribulations. Until Pope Gregory the Great was sent to spread Christianity throughout England, the Anglo- Saxons…
Abraham, also known as Abram is most commonly known for being the Father of the Jewish people. The majority of the information found on Abraham is located in the Old Testament’s Book of Genesis. Other than that, there are no real historical records on the life of Abraham, so the history of his life was…
The positive libidinal feelings of a child to the parent of the opposite sex and hostile or jealous feelings toward the parent of the same sex that may be a source of adult personality disorder when unresolved. It is a pattern of profound emotional ambivalence, a troublesome mixture of love and hate. The Oedipus Complex…
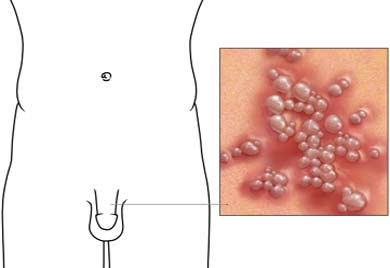
Herpes is a name that is used to describe several types of skin eruptions that are characterized by formation of blisters. The term embraces primarily two distinct disorders, herpes simplex and herpes zoster, both caused by viruses. Together, these “herpes” viruses are estimated to cause more human illnesses than any other group of viruses. Two…
Fetishism is a perversion that is found, primarily in men, in which genital discharge is impossible without the presence of their fetish. A fetish, however can be a variety of things, yet there are three basic types of fetishes: an inanimate object (e.g. women’s clothing, shoes, gloves, underwear), a part of the human body (e.g.…
We would like to think that everything in life is capable, or beyond the brink of reaching perfection. It would be an absolute dream to look upon each day with a positive outlook. We try to establish our lives to the point where this perfection may come true at times, although, it most likely never…
Several examples of physical courage in this excellent novel. One example of courage however insignificant to me or you is Jem’s courage to run-up to the Radley’s house, now to me or you that is nothing but to a young child that has grown up not knowing this person it was a physical challenge to…
The Soviet Space Program, Vostok, was designed to be the first manned spacecraft, and therefore introducing the first man in space. Vostok I succeeded in orbiting the earth on April 12, 1961, and the first man in space was Yuri Gagarin. The vehicle used for the Vostok program consisted of two models. The first model…
Many poems are concerned with a sense of loss or deep sadness at a particular event. The poem ‘Dulce et decorum est’ by Wilfred Owen deals with both loss and deep sadness. Immediately in the poem there are very strong images being used throughout the poem and this shows the sadness from the very start.…
Waterlily is a book based on the events that happen within a particular Dakota Indian family’s life. Not only does it depict the affairs of blue Bird’s life and her family, but it documents the rituals and culture of the Dakota people. With illustrative words and graphic details, the incidences within the story can be…
Rafael Leonidas Trujillo came from a humble family in the Dominican Republic. At the age of 18 he enlisted in the National Army where he quickly rose through the military ranks. In 1930, Trujillo ran for the presidency of the Dominican Republic. His political agenda at the time was to reform education, health care and…
Trainspotting is a captivating story of the random events that occur during a critical time in a group of Scottish junkies’ lives. Irvine Welsh illustrates the confusion, anger, and turmoil many drug addicts are subjected to and what happens once they try to quit. The story is centered around Mark Renton, an ordinary twenty-two-year-old who…
“The Wanderer” is an Anglo-Saxon poem about a lonely wanderer hopelessly alleviating his woes in the posthumous period of his fallen lord. Characteristic of the Anglo-Saxon period, the poem portrays themes of fraternity and loyalty, allegiance, and the tradition of a warrior’s passing. The imagery of the warrior, “the byrny-clad warrior, / The prince in…
“The Trip to Halawa Valley,” is a short story written by Lynne Sharon Schwartz. The story is about a son succeeding and getting married in Hawaii, and the reunification of his divorced parents. The parents become the main focus of the story when the son suggests that they go on a trip to Halawa Valley.…